Exogenous Allergic Alveolitis (EAA)
Exogenous allergic alveolitis (EAA), called hypersensitivity pneumonitis the American literature, is a specific allergic reaction to inhaled organic material. In children this is mostly of bird or fungal origin.
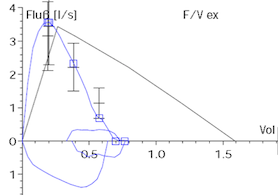 The diagnosis is based on exposure history, dyspnea on exercise, restrictive lung function, HRCT-abnormalities, lymphocytosis on lung lavage and sometimes specific lung biopsy findings.
The diagnosis is based on exposure history, dyspnea on exercise, restrictive lung function, HRCT-abnormalities, lymphocytosis on lung lavage and sometimes specific lung biopsy findings.
Treatment is based on removal of the responsible allergen exposure. This however does not always work, as it is cumbersome, may be incomplete or remains high despite all efforts undertaken. Systemic glucocorticoid treatment is given as standard therapy.
Nevertheless in 25% of cases chronic or severe symptoms develop and these patients cannot be predicted.
During the chILD-EU project, the “Stop-EAA” study was designed and initiated in Germany.
EAA or Exogen Allergic Alveolitis, also called hypersensitivity pneumonitis, is an infrequent chronic pediatric lung disease, which may lead to chronic respiratory failure, if not treated appropriately.
We hypothesized that with appropriate diagnosis, identification of the offending allergen and its removal, only some initial steroid treatment may be necessary. The advantage not to cover too long with steroids would also be not to mask appropriate allergen removal. The role of prednisolone treatment and the long term course has not been followed prospectively in children with EAA.
The primary goal of this work package of the chILD-EU project, to design and initiate a prospective, multicentre, randomized, double-blind, placebo controlled study on hypersensitivity pneumonitis, was achieved. Due to the smaller than expected number of subjects to enrol and lack of long-term funding over many years to guarantee sustained recruitment, the study was stopped.
The very valuable results of the four patients recruited into the trial were analysed in 2020 and are currently in the process of publication.
Specific study aims
- assist allergen identification, verify removal of the allergen exposure and provide secondary exposure prophylaxis
- externally verify removal of the allergen exposure by a home visit
- provide one initial treatment with steroid pulse to help rapid recovery from hypoxemia
- offer a randomized, placebo-controlled medium term treatment with prednisolone
- build up a long term cohort of children for follow up
The study is closed. Participation is no longer possible.
Who can take part in the study?
All newly (or previously diagnosed but not yet appropriately treated) children, adolescents and young adults with EAA
Diagnosis based on the presence of at least 4 of the following findings:
- History of appropriate allergen exposure
- Restrictive lung function (if able to do for age)
- Positive serum precipitins for bird/fungus/rarely other allergens exposed to
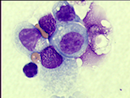
- Lymphocytosis in BAL (> 20% of cells are lymphocytes), if done
- HRCT showing the characteristic nodular, linear or reticular opacities and ground glass pattern with increased attenuation, if done.
- Lung biopsy demonstrating lymphocytic alveolitis, bronchiolitis, and non-caseating histiocytic granulomatas, if done.
- Controlled allergen exposure followed by characteristic reaction, including fever, coughing, restriction on lung function, hypoxemia/desaturation at rest or with exercise, if done.
Diagnosis of EAA confirmed by independent expert panel
Agreement to home visit by independent study physician

