Therapy of malignant thyroid diseases / thyroid cancer
Data on thyroid carcinoma
Thyroid carcinoma is the most frequent tumor derived from endocrine glands. However, with a rate of approx. 1% of all malignant tumors, it is rather rare. In Germany, the annual incidence of thyroid carcinoma is approx. 3-5 new diseases per 100,000 citizens. Mortality is 3-6/1 million citizens/year (~ 0.0005%).Women are approximately twice as often affected as men; incidence rises with advanced age.
Histologically, the so-called differentiated carcinoma (papillary and follicular) are distinguished from the undifferentiated forms (anaplastic) and from medullary carcinoma originating from the para-follicular C cells. The most frequent carcinoma is papillary cancer with approx. 65%, followed by the follicular cancer at 27%. Medullary carcinoma and anaplastic carcinoma are much rarer with less than 5%.
Papillary and follicular thyroid carcinoma generally occur sporadically; however, familiar forms are described for approx. 5% of all cases. In contrast, medullary carcinoma shows familiar predisposition (approx. 25-30%); it is autosomally dominant with almost 100% penetrance in these cases. The reason is a mutation of the so-called RET protooncogene on chromosome 10. In turn half of those patients with the familial form of C cell carcinoma will develop pheochromocytomas (tumors of the adrenal medulla) with so-called multiple-endocrinal adenomatosis (MEA or MEN).
There are no reliably identified risk factors for the development of a thyroid carcinoma, except ionizing radiation (particularly at early ages). Among others, this is e.g. an external radiation exposure during radiation therapy of the neck or incorporation of radio-active iodine. The latter is documented by the increase of the incidence of the thyroid carcinoma in children after the reactor incident in Chernobyl in 1986.
Treatment of the thyroid cancer
The treatment of the thyroid carcinoma at Ludwig Maximilians University at Munich is performed as collaboration between different specialists and is oriented on the guidelines of the German Society of Nuclear Medicine, the German Cancer Society, the German Society of Endocrinology (thyroid division) and the German Society of Surgery. Therapy regularly includes three different pillars: surgery, radio-iodine therapy and life-long substitution with thyroid hormones.
Primary therapy is always surgical, whereby the total thyroidectomy (total removal of the thyroid) is the first choice for most cases of papillary and follicular carcinoma.
Radio-iodine therapy
The next step following surgery is usually an ablative radio-iodine therapy. The aim here is to eliminate (generally benign) residual thyroid tissue and possible remnant tumor cells. The background of radio iodine therapy is that the vast majority of well-differentiated thyroid carcinoma cells have a preserved iodine metabolism.
Radio-iodine therapy reduces the risk of recurrence, most likely due to destruction of occult micro-metastases. As differentiated thyroid tissue is higly iodine avid, an exclusion of metastases in a whole-body scintigraphy with I-131 is reliably possible. In addition the tumor marker thyroglobulin (hTG) has much better diagnostic sensitivity when benigne thyroid tissue is eliminated with I-131. Furthermore, high dose radio-iodine therapy is used to treat local recurrences, lymph node and distant metastases.
Radio-iodine therapy will be perfromed with radio-active I-131, which generally is administered orally as a capsule and reaches the thyroid cells through the blood circulation. The therapy effect is predominantly achieved by the beta radiation during the radioactive disintegration. The beta particles have in the tissue a range of only few millimeters. Thus, a very high radiation exposition in the thyroid tissue can be achieved with minimal affection of the surrounding tissue. The simultaneously generated gamma radiation particles enable the visualization of activity distribution in the so-called post-therapy scintigraphy and are responsible for the necessity of hospitalization for radiation protection purposes.
High TSH levels at the time of therapy are needed for maximized I-131 uptake in residual tissue and tumor cells. Thyroid stimulating hormone (TSH) from the hypophysis stimulates the thyroid cells to a maximum uptake of iodine; targeted TSH values are > 30 µU/ml. This can be achieved by approx. 3 weeks of hormone carence after surgery or by the injection of recombinant TSH for exogenous stimulation.
Radio-iodine therapy is not indicated for patients with medullar or de-differentiated (anaplastic) thyroid carcinoma. Absolute contraindications for radio-iodine therapy are gravidity, therefore exclusion by a pregnancy test is obligatory, and breast-feeding. After radio-iodine therapy, contraception for at least 6-12 months is recommended in order to enable an usually required second application of radio-nuclides, for diagnostic or therapeutic purposes.
Implementation of radio-iodine therapy
Preparation: Due to the required TSH stimulation, a possibly existing thyroid hormone therapy has to be stopped approx. 3-4 weeks prior to radio-iodine therapy. Alternatiely exogenous TSH-stimulation is possible, in which chases patients are allowed to receive thyroid hormones (the latter is the now the usual procedure). Moreover, a strict iodine carence has to be ensured (no application of contrast agents, low-iodine nutrition).
Due to legal regulations, radio-iodine therapy can only be performed as an in-patient treatment in Germany. During the stay in the hospital, the size of the thyroid remnants after surgery is estimated following administration of a very low test activity of iodine-131 (illustration).
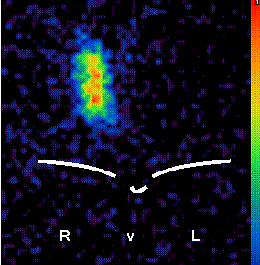 |
| Scintigraphy after thyroid has been almost removed in surgery. |
Bei einem zu großen Schilddrüsenrest muss evtl. in Absprache mit dem Chirurgen eine erneute OP in Betracht gezogen werden. Für die erste Radioiodtherapie werden üblicherweise ca. 3700 MBq (100 mCi) Jod -131 eingesetzt. Am Entlassungstag (üblicherweise 3 Tage nach Gabe der Therapieaktivität) wird mit der im Körper vorhandenen Restaktivität das sog. Posttherapieszintigramm des gesamten Körpers angefertigt (Abbildung). Dieses Szintigramm dient der Überprüfung einer Fernmetastasierung des Schilddrüsenkarzinoms.
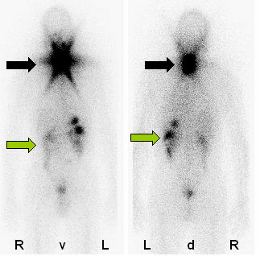 |
| Scintigraphy after ablative radio-iodine therapy shows a activity stored in the area of the rest thyroid (black arrow) and some physiological activity in the intestinal tract (green arrow) but no metastases. |
Likewise, thyroid hormone substitution is started on the day of discharge, where a TSH value of <0,1 or between 0,3 and 1,0 µU/ml is targeted (depending in the stage of disease). This is generally achieved by Levothyroxine doses of approximately 0,5 µg/kg of body weight and day.
Risks and side-effects
Severe side effects of radio-iodine therapy are very rare. Sometimes, temporary inflammation of thyroid and tumor remnants, of salivary glands and gastric mucosa may occur. These side-effects are usually mild and can be treated well with anti-inflammatory procedures (e.g. ice necktie) or anti-inflammatory medications. To prevent damage to the salivary glands, sufficient salivation is necessary, which may be achieved by administration of lemon juice, chewing gum or sour sweets. Moreover, sufficient liquid intake and frequent urination are important.
Permanent damage of the salivary glands is extremely rare and only occurs after multiple high-dose radio-iodine therapies, which may be necessary in andvanced disease. Severe side-effects of radio-iodine therapy occure almost exclusively after high dose therapy of advanced disease and may be: Bone marrow insufficiency, occurring after a total administered activity of more than 30 - 40 GBq (usual initial therapeutic dose 2-3,7 GBq); increased leukemia incidence (approx. 1%), also depending on the dose; Sicca syndrome and lung fibrosis (< 1%) in case of extensive lung metastases. However, there are no hints on an increased frequency of genetic damage; i.e. that there is no increased rate of malformations or other negative effects on pregnancies after radio-iodine therapy.
Follow-up
Although differentiated thyroid cancer, as described, has an excellent prognosis, life-long follow-up is needed since thyroid cancer may develop a so called late recurrence. Therefore, the follow up aims for early discovery of a recurrence or metastases of thyroid cancer. Early detection is of paramaount importance for sucessful treatment.
The most important parameter in the follow-up of differentiated thyroid carcinoma is the so-called thyroglobulin, a protein produced only by thyroid cells. After successful ablation of the remaining thyroid tissue, no thyroglobulin should be measured in the patient’s blood. As a rule, further morphological tests are unnecessary if thyroglobulin levels are below the lower limit. During follow-up the sufficient thyroid hormone substitution is also controlled.
If thyroglobulin levels rise again, it is necessary to search for recurrences or metastases, for example with a diagnostic iodine 131 whole-body scintigraphy (in hypothyroidism) or other imaging methods (e.g. positron emission tomography, computed tomography or magnetic resonance tomography). Depending on these results, adequate therapy options will be chosen, which usually rely on the spectrum of the first therapy e.g a high-dose radio iodine therapy (illustration).
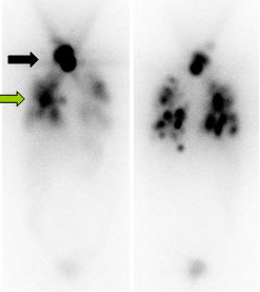 |
| Scintigraphy after first ablative radio-iodine therapy shows a clear iodine uptake in the area of the remnant thyroid tissue (black arrow) and a multiple lung metastases (green arrow). |
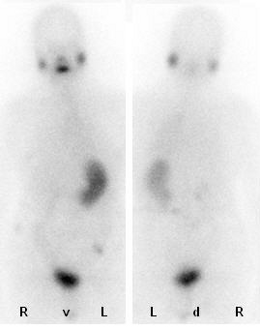 |
|
The scintigraphy shows the same patient after a total of three radio-iodine therapies. There is no proof for remaining metastasis, the tumor marker thyroglobulin is below the lower limit. One can see the physiological iodine uptake in the salivary glanda, intestinal tract and urine bladder. |
